Type A vs Type B ADR Classifier
How to Use This Tool
Based on the characteristics described in the article, identify whether the adverse drug reaction described is Type A or Type B.
- Type A: Predictable, dose-dependent, common
- Type B: Unpredictable, not dose-dependent, rare but often severe
Case Scenario
When you take a medication, you expect it to help - not hurt. But sometimes, drugs cause unexpected or unwanted side effects. Not all of these reactions are the same. Some are common and predictable. Others are rare, dangerous, and totally unpredictable. Understanding the difference between Type A and Type B adverse drug reactions (ADRs) can make all the difference in avoiding harm, spotting trouble early, and knowing when to act.
Here’s the reality: about 85 to 90% of all drug side effects fall into Type A. That means most reactions you’ve probably experienced - like nausea from antibiotics or dizziness from blood pressure meds - are not surprises. They’re built into how the drug works. The other 10 to 15%? Those are Type B. These are the ones that catch doctors off guard. They can be life-threatening, happen to just one person out of 10,000, and have no clear link to the dose you took. This isn’t just theory. It’s happening in hospitals, clinics, and homes every day.
What Are Type A Adverse Drug Reactions?
Type A reactions are the predictable ones. They happen because of how the drug is supposed to work - just taken too far. Think of them as an extension of the drug’s intended effect, turned up too loud.
- Example: Taking too much acetaminophen? You risk liver damage. That’s not a fluke - it’s direct toxicity from exceeding the safe dose.
- Example: Starting a beta-blocker for high blood pressure? You might feel tired or have a slow heart rate. That’s the drug doing its job… too well.
- Example: NSAIDs like ibuprofen? Around 15-30% of users get stomach upset. That’s because these drugs block protective enzymes in the gut. It’s known. It’s expected.
These reactions are dose-dependent. Take more, get worse. Take less, it fades. That’s why doctors adjust doses. That’s why you’re told not to crush pills or double up. Type A reactions make up the bulk of side effects you see in prescribing guides. They’re why drug labels have warnings like “may cause drowsiness” or “avoid alcohol.”
They’re also why hospital pharmacies have automated alerts. If a patient’s kidney function is low and the system sees a dose of a kidney-cleared drug, it flags it. That’s Type A prediction in action.
What Are Type B Adverse Drug Reactions?
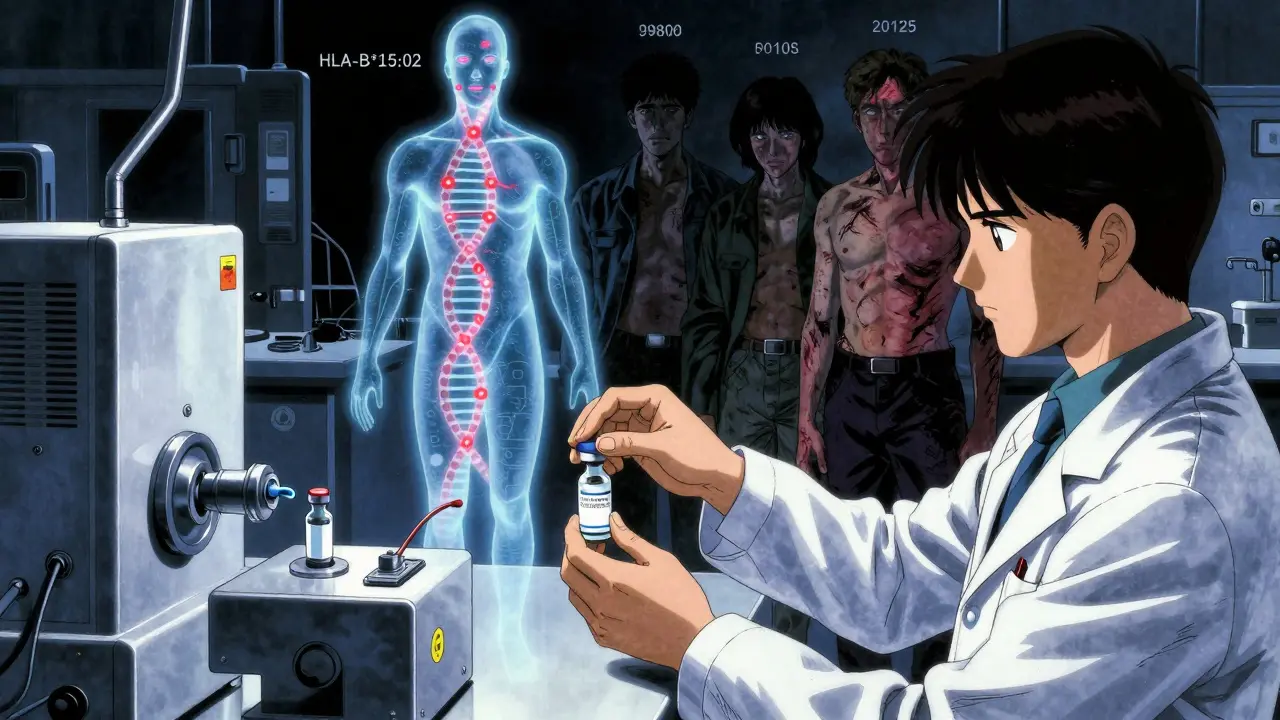
Type B reactions are the wild cards. They’re not related to the drug’s main action. They’re not dose-dependent. And they’re often immune-driven or tied to your unique biology.
- Example: A patient takes penicillin and goes into anaphylaxis - swelling, trouble breathing, drop in blood pressure. This happens in 0.01% to 0.05% of courses. No amount of dose adjustment helps. You can’t predict it by age, weight, or kidney function.
- Example: Sulfonamide antibiotics trigger Stevens-Johnson syndrome - a terrifying skin reaction that peels off like a burn. It affects roughly 1 to 6 people per million prescriptions. It’s not about the dose. It’s about your genes.
- Example: A healthy young adult takes a common anticonvulsant like carbamazepine and develops a severe rash. Turns out they carry the HLA-B*15:02 gene. That’s a genetic red flag. This used to be called “idiosyncratic.” Now we know it’s genetic.
Type B reactions are rare - but deadly. They cause 30% of all ADR-related hospitalizations. Even though they’re only 10-15% of all reactions, they’re responsible for most drug withdrawals from the market. Why? Because you can’t dose your way out of them. You can’t warn patients with a general label. You need to know who’s at risk - and that’s where things get complicated.
The Six-Type Classification System: Beyond A and B
The A/B system is useful, but it’s not enough. In 2023, most serious ADR reporting in Europe and the U.S. uses a six-type system that adds depth.
- Type C - Chronic effects from long-term use. Think corticosteroids. If you take more than 20mg of prednisone daily for over three weeks, you have a 20-30% chance of adrenal suppression. This isn’t sudden. It builds over time.
- Type D - Delayed reactions. The classic example? Diethylstilbestrol (DES). A drug given to pregnant women in the 1950s to prevent miscarriage. Decades later, their daughters developed a rare vaginal cancer. The damage didn’t show up until adulthood.
- Type E - Withdrawal reactions. Stop opioids? 80-90% of dependent patients get withdrawal symptoms within 12-30 hours. It’s not an allergic reaction. It’s the body’s physical dependence kicking in.
- Type F - Therapeutic failure. This one’s often missed. Rifampin, an antibiotic, speeds up how your liver breaks down oral contraceptives. Result? Birth control fails in 5-10% of cases. The drug worked - just not the way you thought.
These categories help explain reactions that don’t fit neatly into “dose-related” or “unpredictable.” A doctor who only thinks in Type A and Type B might miss a Type F interaction - and a patient could get pregnant because no one checked for drug interference.

Why the Classification Matters
It’s not just academic. Knowing the difference changes how you treat.
If it’s Type A - adjust the dose. Monitor. Educate. Maybe switch to a different drug in the same class.
If it’s Type B - stop the drug. Full stop. No second chances. Don’t try a lower dose. Don’t “see how it goes.” You’re not dealing with a side effect. You’re dealing with a potential death sentence.
And here’s the twist: modern science is blurring the lines. A reaction once labeled Type B - like carbamazepine-induced rash - is now known to be tied to specific genes. That means it’s not random anymore. We can test for it. We can prevent it. That’s why the FDA and WHO are pushing for pharmacogenomic screening before prescribing certain drugs.
But not every hospital can do genetic testing. Not every doctor has time. So the A/B framework still holds. It’s the first filter. The next step? Knowing when to dig deeper.
Real-World Challenges
Doctors don’t always agree on classification. A 2022 survey of over 1,200 physicians found that 67% struggled with ambiguous cases. Is a low sodium level from carbamazepine a Type A (dose-related) or Type B (idiosyncratic) reaction? Studies show it’s dose-dependent - so Type A. But if it only happens in people with a certain gene? Now it’s both.
And here’s the silent problem: Type F reactions - therapeutic failures - are missed in 25% of cases. A woman on birth control gets a prescription for rifampin. No one tells her. She gets pregnant. That’s not a side effect. That’s a system failure.
Meanwhile, Type B reactions like drug-induced liver injury or anaphylaxis are underreported. Patients don’t connect the dots. Clinicians assume it’s a virus or a flare-up. Only 65% of serious ADRs can be definitively classified - even with today’s tools.
What You Can Do
If you’re a patient:
- Know your meds. Ask: “What are the common side effects? What’s the rare but dangerous one?”
- Report anything unusual - even if it seems minor. A rash, dizziness, or unexplained fatigue could be the first sign.
- If you’ve had a bad reaction before, tell every new doctor. Write it down. Include the drug name and what happened.
If you’re a caregiver or healthcare provider:
- Use the six-type system. Don’t stop at A and B.
- Check for drug interactions. Especially with antibiotics, antifungals, and seizure meds.
- Consider pharmacogenomics for high-risk drugs - like abacavir, carbamazepine, or clopidogrel.
- Use automated alerts. Most hospital systems now flag Type A risks. Don’t ignore them.
The goal isn’t to scare you. It’s to empower you. Most drug reactions are harmless and manageable. But the ones that aren’t? They often come without warning. Understanding the difference between Type A and Type B gives you a better chance to spot trouble before it’s too late.
What’s Next?
By 2027, experts predict 60% of what we now call Type B reactions will have known genetic causes. That means “idiosyncratic” will become “predictable.” We’ll test before we treat. We’ll prevent before we harm.
The World Health Organization is testing a new system that links genetic data directly to ADR alerts. The FDA is updating its reporting rules to require mechanistic details for every serious Type B reaction. And by 2025, the six-type classification will be the global standard.
But for now? The A/B split still saves lives. It’s simple. It’s clear. And when used right - with awareness of the deeper layers - it’s powerful.
Steven Pam
February 23, 2026 AT 09:37Type A reactions are the boring stuff we all know about-nausea, dizziness, dry mouth. But Type B? That’s where things get wild. I had a friend go into anaphylaxis from amoxicillin. No prior history. No warning. Just… gone. One minute laughing, next minute in the ER. That’s the nightmare no drug label prepares you for.
And honestly? We treat Type A like it’s the whole story. But Type B is the silent killer. It doesn’t care how careful you are. It doesn’t care if you’re young, healthy, or read every warning. It just… happens. And when it does? You pray you get to the hospital in time.
That’s why I’m all for genetic screening. If we can test for HLA-B*15:02 before giving carbamazepine, why aren’t we doing it everywhere? It’s not expensive. It’s not complicated. It’s just not prioritized.
Doctors don’t have time. Pharmacies don’t flag it. Patients don’t know to ask. So we keep playing Russian roulette with our meds.
I’m not saying stop taking drugs. I’m saying: know your risk. Push for testing. Demand better. Because someone’s kid could be next.
Natanya Green
February 23, 2026 AT 23:47OMG, this post is SO important!!! I literally just had a Type B reaction last year-rash, fever, my whole body felt like it was on fire!! I thought it was the flu, but nooo-it was sulfa antibiotics!! My doctor didn’t even think to connect it!!
Now I carry a card in my wallet that says: ‘ALLERGY: SULFONAMIDES-DO NOT ADMINISTER.’ I’m basically a medical superhero now. 😎
Also, why isn’t this on every prescription bottle?? Like, hello??
Timothy Haroutunian
February 25, 2026 AT 17:08Let’s be real-the Type A/B distinction is a lazy oversimplification. It’s a textbook relic. The real issue is that medicine still operates on population averages while ignoring individual biology. You don’t need a six-type system-you need a personalized one. Every person’s metabolism, microbiome, and epigenetics should be factored in before a single pill is prescribed.
And yet, we still rely on ‘dose-dependent’ and ‘idiosyncratic’ as if those are scientific categories and not just placeholders for ignorance.
The fact that we’re still using 1970s terminology in 2025 is embarrassing. Pharmacogenomics isn’t the future-it’s the present. And we’re still pretending it’s optional.
Stop treating patients like statistical noise. Start treating them like unique biological systems.
Erin Pinheiro
February 26, 2026 AT 08:24i read this and was like… wait so like… if i got a rash from penicillin as a kid does that mean i have type b? or do i just suck at being healthy??
also why do they always say ‘rare’ but then list numbers like 1 in a million?? like… if 100 people get it every year in the us, is that still rare??
and what about type f?? i had a baby after being on birth control and my doc prescribed me rifampin for tb and never said a word. i’m not mad, just confused. why is this not a thing we talk about??
Michael FItzpatrick
February 27, 2026 AT 22:03What’s beautiful here is how this framework reveals medicine’s evolution-from guesswork to precision. Type A was the era of ‘more is worse.’ Type B was the era of ‘why did this happen to her?’ Now we’re entering the era of ‘we can see it coming.’
Imagine a world where your EHR doesn’t just show your blood pressure-it shows your HLA profile. Where your pharmacist says, ‘Based on your genetics, we’re avoiding this class of drugs.’ That’s not sci-fi. It’s happening in academic centers right now.
The real revolution isn’t in the drugs. It’s in the data. And the people who still treat ADRs as ‘bad luck’ are the ones holding medicine back.
Let’s not wait for the next tragedy. Let’s build the systems that prevent them.
Brandice Valentino
March 1, 2026 AT 08:34Ugh, I’m so tired of this ‘Type A/B’ nonsense. It’s like calling all dogs ‘barks’ and ‘doesn’t bark.’ So reductive. And don’t even get me started on the ‘six-type’ system-sounds like a grad student’s thesis with too much caffeine.
Real talk: if you’re taking meds and something weird happens, STOP. Call your doctor. Don’t wait for a classification system to validate your suffering.
Also, why are we still using ‘idiosyncratic’? That word is just a fancy way of saying ‘we don’t know.’ And we should stop pretending we do.
Larry Zerpa
March 1, 2026 AT 20:23Let’s cut the fluff. Type B reactions are just the medical system’s way of saying, ‘We don’t understand your body.’ It’s not a category-it’s a cop-out. And now we’re adding Type C through F like we’re building a Dungeons & Dragons spellbook.
Here’s what actually matters: if a drug kills you, it shouldn’t be on the market. Period. Stop hiding behind classifications. If it’s dangerous, remove it. If it’s risky, test for it. If it’s ignored, sue someone.
And for god’s sake, stop telling patients to ‘report anything unusual.’ That’s like telling someone to ‘call if you feel weird’ after a car crash. We need mandatory reporting. We need liability. We need consequences.
This isn’t education. It’s damage control.
Christopher Brown
March 3, 2026 AT 15:51America’s healthcare system is a joke. We have the tech to prevent Type B reactions. We have the data. But we won’t fund genetic screening because it’s ‘too expensive’-until someone dies and then we pay $200k in ER bills.
Meanwhile, China and Germany are already mandating HLA testing for carbamazepine. We’re still arguing over whether Type F counts as a ‘side effect.’
Stop pretending we’re leaders in medicine. We’re lagging. And we’re paying for it with lives.
Kenzie Goode
March 4, 2026 AT 20:16This post gave me chills. I’ve been a nurse for 18 years, and I’ve seen Type B reactions take people in seconds. No warning. No rhyme or reason.
But here’s what I’ve learned: patients who speak up survive. The ones who say, ‘I had a rash last time’-they live. The ones who stay quiet? They don’t.
I wish every patient had this info. Not because it’s scary-but because knowledge is armor.
Please, if you’re reading this: write down your reactions. Tell every provider. Even if they roll their eyes. Even if it’s been 10 years.
Your life might depend on it.
Dominic Punch
March 5, 2026 AT 21:29As someone who works in UK primary care, I’ve seen Type F failures daily. A woman on the pill gets a course of rifampin for TB. No interaction check. No warning. She gets pregnant. Then she blames herself.
We need automated alerts in every e-prescribing system. We need mandatory patient education sheets. We need pharmacists to be the final gatekeepers, not afterthoughts.
And yes-we need pharmacogenomics. But we also need to stop treating patients like passive recipients of care. They’re the first line of defense. Equip them.
Simple. Effective. Necessary.
Brooke Exley
March 5, 2026 AT 22:44I love how this breaks it down without jargon. It’s like a safety manual for your body.
One thing I’d add: Type A reactions? They’re your body’s way of saying, ‘Hey, maybe this isn’t for you.’ Listen. Don’t just push through the dizziness or nausea.
And Type B? If you’ve ever had a strange reaction-even if it was years ago-don’t brush it off. That’s your body’s whisper. Pay attention.
You know your body better than any algorithm. Trust it. And tell someone.
That’s how we save lives.
Jacob Carthy
March 6, 2026 AT 08:33Type A is just side effects Type B is just bad luck
end of story